原子力显微镜
AFM配件
应用
联系我们
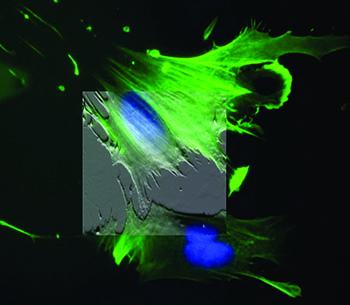
原子力显微镜(AFM)是细胞生物学研究的重要仪器,它可以提供活细胞、非固定细胞的三维形貌数据。然而,在细胞生物学研究中主要的优势是它能够在近生理条件下(例如:在培养基和37摄氏度条件下)提供准确、量化的机械测量。同时,利用力成像模式和基于AFM的微流变技术,可以对细胞或基质的弹性和粘弹性响应进行常规测量。测量的细胞模量可以是未被改变的细胞、不同发育状态的细胞、变异细胞、染病细胞,或者对某些刺激(例如:药物或机械压力)作出反应的细胞。由于细胞外基质(ECM)在一些过程中(例如:细胞的分化、命运走向、信号、基因转录、癌症、心血管疾病和细胞死亡等)发挥着作用,测量基质的模量和细胞微环境是非常重要的。
如果集成了倒置的显微镜(例如:荧光、共焦和TIRF等),两种成像模式的数据可以结合在一起,利用的AFM形貌成像,将荧光标记的结构与形貌图相关联。这些元件可用来引导AFM的探针针尖进入细胞的特定区域,这对于那些不容易成像的细胞来说是非常关键的。此外,还可以用来为细胞提供机械刺激,并可以用来记录相关的反应(例如:离子处理和膜电位变化等),以了解活细胞和组织中的机械传导。
咨询AFM领域的专家"Peptide density targets and impedes triple negative breast cancer metastasis," D. Liu, P. Guo, C. McCarthy, B. Wang, Y. Tao, and D. Auguste, Nat. Commun. 9, 2612 (2018). https://doi.org/10.1038/s41467-018-05035-5
"Direct micropatterning of extracellular matrix proteins on functionalized polyacrylamide hydrogels shows geometric regulation of cell–cell junctions," B. Sarker, C. Walter, and A. Pathak, ACS Biomater. Sci. Eng. 4, 2340 (2018). https://doi.org/10.1021/acsbiomaterials.8b00331
"Measuring nanoscale viscoelastic parameters of cells directly from force-displacement curves," Y. M. Efremov, W.-H. Wang, S. D. Hardy, R. L. Geahlen, and A. Raman, Sci. Rep. 7, 1541 (2017). https://doi.org/10.1038/s41598-017-01784-3
"3D cell bioprinting of self-assembling peptide-based hydrogels," B. Raphael, T. Khalil, V. L. Workman, A. Smith, C. P. Brown, C. Streuli, A. Saiani, and M. Domingos, Mater. Lett. 190, 103 (2017). https://doi.org/10.1016/j.matlet.2016.12.127
"T cell activation requires force generation," K. H. Hu and M. J. Butte, J. Cell Biol. 213, 535 (2016). https://doi.org/10.1083/jcb.201511053
"Mechanical heterogeneities in the subendothelial matrix develop with age and decrease with exercise," J. C. Kohn, A. Chen, S. Cheng, D. R. Kowal, M. R. King, and C. A. Reinhart-King, J. Biomech. 49, 1447 (2016). https://doi.org/10.1016/j.jbiomech.2016.03.016
"Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration," I. Acerbi, L. Cassereau, I. Dean, Q. Shi, A. Au, C. Park, Y. Y. Chen, J. Liphardt, E. S. Hwang, and V. M. Weaver, Integr. Biol. 7, 1120 (2015). https://doi.org/10.1039/c5ib00040h
"Nanomechanics of cells and biomaterials studied by atomic force microscopy," J. I. Kilpatrick, I. Revenko, and B. J. Rodriguez, Adv. Healthcare Mater. 4, 2456 (2015). https://doi.org/10.1002/adhm.201500229
"Tissue mechanics modulate microRNA-dependent PTEN expression to regulate malignant progression," J. K. Mouw, Y. Yui, L. Damiano, R. O. Bainer, J. N. Lakins, I. Acerbi, G. Ou, A. C. Wijekoon, K. R. Levental, P. M. Gilbert, E. S. Hwang, Y.-Y. Chen, and V. M. Weaver, Nat. Med. 20, 360 (2014). https://doi.org/10.1038/nm.3497
"Atomic force microscopy-based microrheology reveals significant differences in the viscoelastic response between malign and benign cell lines," J. Rother, H. Nöding, I. Mey, and A. Janshoff, Open Biol. 4, 140046 (2014). https://doi.org/10.1098/rsob.140046
"A physical sciences network characterization of non-tumorigenic and metastatic cells," The Physical Sciences – Oncology Centers Network (D. B. Agus et al.), Sci. Rep. 3, 1449 (2013). https://doi.org/10.1038/srep01449
"Comparison of the viscoelastic properties of cells from different kidney cancer phenotypes measured with atomic force microscopy," L. M. Rebelo, J. S. de Sousa, J. Mendes Filho, and M. Radmacher, 24, 055102 (2013). https://doi.org/10.1088/0957-4484/24/5/055102
"Elasticity maps of living neurons measured by combined fluorescence and atomic force microscopy," E. Spedden, J. D. White, E. N. Naumova, D. L. Kaplan, and C. Staii, Biophys. J. 103, 868 (2012). https://doi.org/10.1016/j.bpj.2012.08.005
"Cell stiffness is a biomarker of the metastatic potential of ovarian cancer cells," W. Xu, R. Mezencev, B. Kim, L. Wang, J. McDonald, and T. Sulchek, PLoS One 7, e46609 (2012). https://doi.org/10.1371/journal.pone.0046609
"In situ force mapping of mammary gland transformation," J. I. Lopez, I. Kang, W.-K. You, D. M. McDonald, and V. M. Weaver, Integr. Biol. 3, 910 (2011). https://doi.org/10.1039/c1ib00043h
"Mapping nanomechanical properties of live cells using multi-harmonic atomic force microscopy," A. Raman, S. Trigueros, A. Cartagena, A. P. Z. Stevenson, M. Susilo, E. Nauman, and S. A. Contera, Nat. Nanotechnol. 6, 809 (2011). https://doi.org/10.1038/nnano.2011.186
"Detection of single-molecule H2O2 signalling from epidermal growth factor receptor using fluorescent single-walled carbon nanotubes," H. Jin, D. A. Heller, M. Kalbacova, J.-H. Kim, J. Zhang, A. A. Boghossian, N. Maheshri, and M. S. Strano, Nat. Nanotechnol. 5, 302 (2010). https://doi.org/10.1038/nnano.2010.24
"Nanotopography-induced changes in focal adhesions, cytoskeletal organization, and mechanical properties of human mesenchymal stem cells," E. K. Yim, E. M. Darling, K. Kulangara, F. Guilak, and K. W. Leong, Biomaterials 31, 1299 (2010). https://doi.org10.1016/j.biomaterials.2009.10.037
"Impact of actin rearrangement and degranulation on the membrane structure of primary mast cells: A combined atomic force and laser scanning confocal microscopy investigation," Z. Deng, T. Zink, H. Chen, D. Walters, F. Liu, and G. Liu, Biophys. J. 96, 1629 (2009). https://doi.org/10.1016/j.bpj.2008.11.015
"Control of myocyte remodeling in vitro with engineered substrates," N. A. Geisse, S. P. Sheehy, and K. K. Parker, In Vitro Cell. Dev. Biol.–Animal 45, 343 (2009). https://doi.org/10.1007/s11626-009-9182-9
"Intermediate filament-like proteins in bacteria and a cytoskeletal function in Streptomyces," S. Bagchi, H. Tomenius, L. M. Belova, and N. Ausmees, Mol. Microbiol. 70, 1037 (2008). https://doi.org/10.1111/j.1365-2958.2008.06473.x
"A cell nanoinjector based on carbon nanotubes," X. Chen, A. Kis, A. Zettl, and C. R. Bertozzi, Proc. Natl. Acad. Sci. U. S. A. 104, 8218 (2007). https://doi.org/10.1073/.0700567104
"Matrix elasticity directs stem cell lineage specification," A. J. Engler, S. Sen, H. L. Sweeney, and D. E. Discher, Cell 126, 677 (2006). https://doi.org/10.1016/j.cell.2006.06.044
"Indentation and adhesive probing of a cell membrane with : Theoretical model and experiments," S. Sen, S. Subramanian, and D. E. Discher, Biophys. J. 89, 3203 (2005). https://doi.org/10.1529/biophysj.105.063826
"Myotubes differentiate optimally on substrates with tissue-like stiffness: pathological implications for soft or stiff microenvironments," A. J. Engler, M. A. Griffin, S. Sen, C. G. Bönnemann, H. L. Sweeney, and D. E. Discher, J. Cell Biol. 166, 877 (2004). https://doi.org/10.1083/jcb.200405004
 公安机关备案号31010402003473
公安机关备案号31010402003473